 
It is an in vitro alternative to the traditional hybridoma-based technology for creating monoclonal antibodies. However, the main disadvantage with this technology is the difficulty of producing large quantities of stable human monoclonal antibodies, due to the absence of suitable myeloma cell lines.Ī recombinant antibody is a synthetic antibody or antibody fragment that is generated by recombinant DNA technology. If intended for therapeutic use, the antibody must undergo humanization, wherein parts of the protein sequence - usually the constant regions - are replaced with the human equivalent otherwise, it may elicit an unintended immune response in humans. Also, hybridomas are sometimes difficult and expensive to maintain in culture. More than 99% of the cells do not survive the fusion process, thus reducing the range of useful antibodies that can be produced against an antigen. Alternatively, monoclonal antibodies can be produced in vivo by injecting hybridoma cells into the peritoneal cavity of an animal and harvesting antibodies from the resulting fluid (ascites).Īlthough hybridoma antibody production is a well-established method of producing antibody, there are several drawbacks. The candidate cell lines are cultured, and monoclonal antibodies are purified from the supernatant.The hybridomas are subjected to multiple rounds of screening and selection to identify ones that produce the best mAbs for the intended downstream application.These B cells are isolated from the spleen of the host animal and fused with tumor cells to generate hybridomas.A host animal is immunized with the antigen to elicit an immune response and initiate the process of B-cell maturation.An immunogenic antigen is developed and optimized.Hybridoma generation is a five-step process that takes advantage of a host animal's natural ability to generate functional, highly specific, high-affinity antibodies (Figure 1). Each hybridoma constitutively expresses a large amount of one specific monoclonal antibody, and preferred cell lines can be cryopreserved for long-lasting mAb production. Hybridomas are cells formed via fusion between a short-lived antibody-producing B cell and an immortal myeloma cell. Hybridoma development is one of the more traditional methods used to generate monoclonal antibodies, enabling the production of highly sensitive binders. Difficult and expensive to maintain in culture.Produces highly pure and specific antibodies. 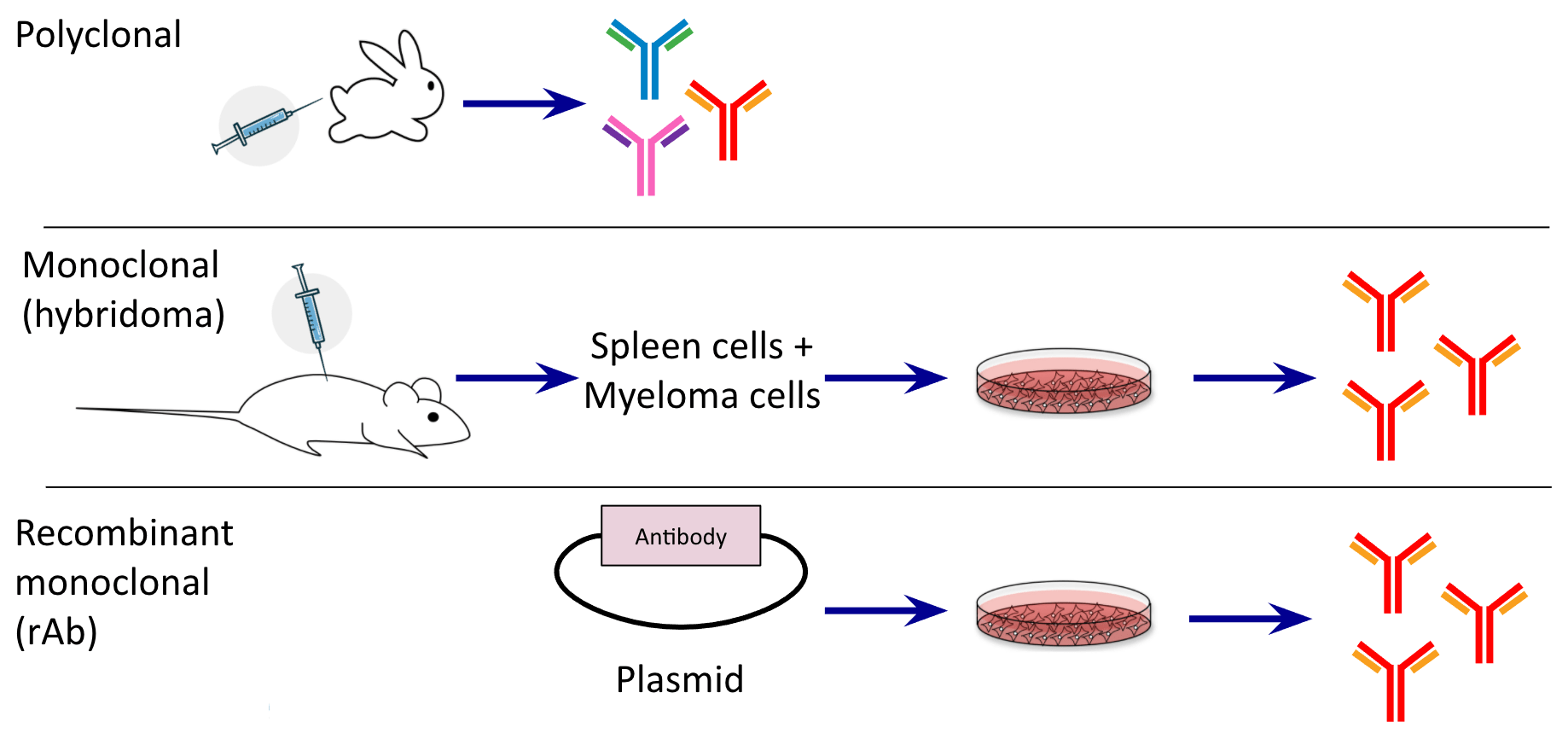
To help you decide which method of antibody production is the most appropriate for your needs, we will look at the process, benefits, and challenges of each one. However, increased commercial demands and quality requirements have caused many companies to explore recombinant antibody production as an alternative. Hybridoma technology, which uses an animal-based approach, has long been the industry standard for monoclonal antibody production. The amount of mAb needed and the importance of factors such as cost, turnaround time, and regulatory compliance depends on the purpose. Decreasing immunogenicity in humans, improving bioavailability, optimizing affinity and antigen-binding specificity, and other advances in protein engineering have contributed to better profiles for therapeutic antibodies. In recent years, technological improvements in antibody design have significantly expanded mAb development. They have various applications in the diagnosis and treatment of a wide range of diseases including cancers, autoimmune disorders, and sexually transmitted infections. Monoclonal antibodies (mAbs), composed of unique pairs of heavy and light chains, have been widely used by researchers to target antigens with high specificity. GENEWIZ Multiomics & Synthesis SolutionsĪntibodies are specialized proteins produced by the immune system that bind and neutralize foreign invaders such as viruses, bacteria, fungi, or parasites.Discovery, Compound Management & Biologics. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
